GRAS: Generally Recognized As Safe
How the FDA Approves Food Additives without a Formal Process

The year was 1958.
Sputnik 1 & Sputnik II crash and burn.
The hula hoop was invented.
Bobby Fischer won the U.S. Chess Championship.
Elvis Presley joined the Army.
Pizza Hut was founded.
Instant noodles hit the market for the first time.
And this is the magic year the FDA picked to declare all food commonly served to be “generally recognized as safe” unless tested and proven to be otherwise.
If people were eating it in 1958 and are still eating it now, well…there you go. Of course, they did not wear seatbelts, thought cigarettes were the coolest ever, and going out with wet hair could cause a cold.
What is “GRAS”?
“Generally Recognized as Safe” is the FDA designation for food additives that do not need to go through the formal approval process because they have either
1) been in use since 1958 (and this arbitrary rule was determined in 1998)
2) have been recognized by “qualified experts” as safe for the additive’s intended purpose…or
3) are not technically defined as a food additive
So we are defining the thing-by-the-thing. Let me clarify on point #3…a food additive is generally recognized as safe if it is not considered a food additive. Clear as mud? This is the FDA’s official definition not mine. I couldn’t make this stuff up. Note that I underline everything that begets more questions for me, and yes, I question everything.
I could write a 5 Part Series on this:
- How do they define the word “additive?”
- Exactly who is a “qualified expert?”
- How can you intentionally add something as an additive that has otherwise been “excluded from the definition of a food additive?”
- So they base this on published studies–what are their criteria for this, since they allow this to be corroborated by unpublished studies and “other sources”? And who pays for these studies? You do not see the American Lung Association paying for a study on the benefits of smoking do you?
- What is a “substantial history” and does it include one where everyone is dying from diabetes, heart disease, and autoimmune diseases? Does it include one with an inexplicable explosion of ADD, ADHD, Depression, Autism, Sensory Processing Disorders (or Sensory Integration Disorders) and other neurological and psychological disabilities?
Hmmm, just wondering.
But I won’t write that 5 Part Series. It is probably enough to know that there are many unanswered questions and more circular thinking than in a room full of football commentators.
You also have to read this fine print:
(1)In addition, a substance that is used in accordance with a sanction granted prior to September 6, 1958 is not subject to premarket approval.
What I called an arbitrary rule had a pragmatic purpose. When the FDA had newly grasped powers to determine what additives were to be, they expected an onslaught of everyday basic items by manufacturers anxious to keep products on market. Take a look down the spice aisle at the grocery store…that is just the tip of what would need to be itemized. And so, in an effort to curb this flood for approval, they needed to blanket-approve many items at once. They also knew they would miss a few along the way that people have been eating for generations, and so they offered another solution…GRAS.
Basically, the process is this: anyone (yes, any mammal with an internet connection) can file a GRAS status with the FDA about a specific substance which includes the substance’ intended use and other data.
Nearly a total of 500 have been formally filed since 1998, but thousands more were grandfathered in at the time the 1998 policy was enacted from previous approvals. The FDA claims one of three results:
- The FDA does not question the item’s status as GRAS
- The FDA says that there is not enough information to make a determination
- The FDA says that the notifier (the person who filed for GRAS status) has withdrawn the GRAS notice
Should we be concerned? We are busy people without chemistry degrees, and the FDA is looking out for our health and safety. Maybe we should just trust them, eh?
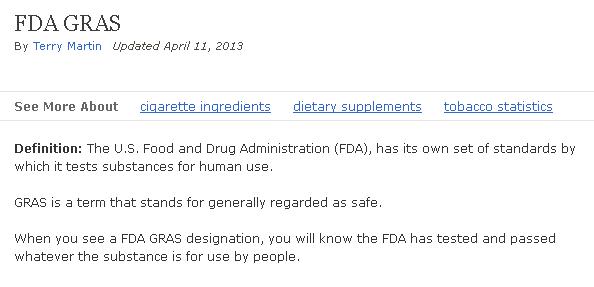
Some people do trust the FDA implicitly. Check out this misinformation from About.com:
Boy, if this isn’t the “because we said so” argument. It didn’t work on you when you were 12 years old. From the documentation I have already provided, I think you can clearly see that the FDA has NOT tested what they declare GRAS, by the FDA’s own definition of GRAS! My point is this–research this for yourself but be sure to cross-examine!
If you’re still drinking the GMO koolaid with Red Dye 40, so to speak…
Three words for you: Brominated Vegetable Oil.

Its’ use dates back to the 1930s you know, it is GRAS, and 10% of the bottled drinks in our country contain BVO (source cited: Stephanie). It is banned in Europe and Japan, and the Mayo Clinic has a thing or two to say about it. Of the 10,000 chemicals permitted in food, I just named one. Do you want me to keep going?
9,999 to go…
Oh, and one final thought: People have been drinking raw milk for millennia, but that is GRAU (Generally Regarded As Unlawful)
Eat real food,
Chaya
Be sure to check out our store, chock full of tools and non-gmo spices to help you navigate the murky waters of feeding your family in unhealthy & financially tough times.
Proviso:
Nothing in this blog constitutes medical or legal advice. You should consult your own physician before making any dietary changes. Statements in this blog may or may not be congruent with current USDA or FDA guidance.
Photo Credits:
- Hula Hoop: cc
- The Definition of GRAS can be found on the FDA website here.
- Cat on computer: Vicki’s Pics via photopin cc
- The About.com misinformation can be found here.
- Bromine Water: maticulous via photopin cc
Sources:
- FDA. Generally Recognized As Safe. http://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/default.htm
- GRAS Notice Inventory, FDA Website: http://www.accessdata.fda.gov/scripts/fcn/fcnNavigation.cfm?rpt=grasListing&displayAll=true
- Stephanie, S. (n.d.). Retrieved from http://www.nytimes.com/2012/12/13/business/another-look-at-a-drink-ingredient-brominated-vegetable-oil.html?pagewanted=all&_r=0
Further Reading:
You will learn more from this than anything else I have found; they are toxicologists with a long history or working with food manufacturers and the FDA in gaining GRAS status: http://www.gras-associates.com/What_is_GRAS_Status_.html
Here is a good article, but I was not able to substantiate all of his facts through other research: http://www.huffingtonpost.com/robert-lustig-md/fda-food-additives_b_3384629.html
DuPont just had some probiotic bacterial strains accepted with GRAS status in November 2013: http://newhope360.com/breaking-news/dupont-probiotics-obtain-fda-gras